At Bryter Labs, we are always experimenting with new techniques for market research. We’re passionate about providing the most insightful answers to our clients’ questions, and often this means using advanced and innovative methods.
MCDA is a highly flexible and powerful analytical approach that unpicks multi-level decision-making. It works by breaking down the available options into their component parts, and then comparing these parts. This structures the decision-making process. Ideal for understanding prescription decision-making. It can also be used to understand the features of a product that are most important to customers, and the features that are driving a high product rating.
For example, when considering which treatment to prescribe, doctors consider features such as efficacy, safety, and dosing. With an MCDA, doctors rate the importance of these features, and then rate each of the treatments on each feature.
Which features are more important? How high does a treatment need to score on each feature in order to be considered? What level of safety is acceptable? MCDA can give us numerical answers to these questions, without requiring the large sample size of a conjoint.
In a normal rating question, doctors are inclined to rate products based on past experience. Their responses can often be biased by factors such as how comfortable they are with a product. Breaking down the rating using an MCDA allowed us to gain more objective insight into what they really thought about each product.
 MCDA can be used in both quantitative and qualitative research. It is quick and works with small sample sizes, making it cost effective for research into rare or orphaned conditions.
MCDA can be used in both quantitative and qualitative research. It is quick and works with small sample sizes, making it cost effective for research into rare or orphaned conditions.
These advantages also make MCDA an attractive method for assessing pharmaceutical products in the early stages of development. MCDA can help to drive the success of a new product by providing an understanding of which product features marketing should focus on.
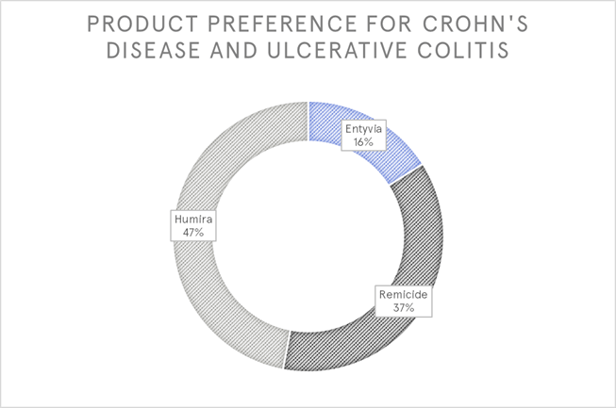
We used the MCDA approach to evaluate three products for Crohn’s disease and ulcerative colitis: Humira, Remicade and Entyvio.
Participants in the survey were asked their preferred of the 3 products to be assessed.
 MCDA required respondents to indicate the relative importance product features that included: Duration of effect, mode/frequency of administration and time to administer.
MCDA required respondents to indicate the relative importance product features that included: Duration of effect, mode/frequency of administration and time to administer.
Here, the MCDA provided a unique insight into doctors’ product ratings. Preference obtained from a simple rating scale were different to those revealed by the MCDA.
Entyvio, a newer product with a different mechanism of action to the other products, came third in the simple rating, but second in the MCDA. This was driven by its longer-lasting effect. Coupling this aspect with efficacy will help Takeda be in tune with physicians' own decision-making process to win market share from the older products that doctors have greater experience and are more comfortable.
In a nutshell, MCDA makes it possible to structure the process of product rating. This structured data can reveal a range of insights that a simple rating cannot. Here at Bryter we are now using MCDA as one of a number of innovative methods in our toolkit. To find out more about this and other advanced analytical techniques we employ get in touch with noemi.hahn@bryter-us.com
.jpg?width=1200&height=600&name=architecture-black-and-white-challenge-chance-277593%20(1).jpg)




